- Blog
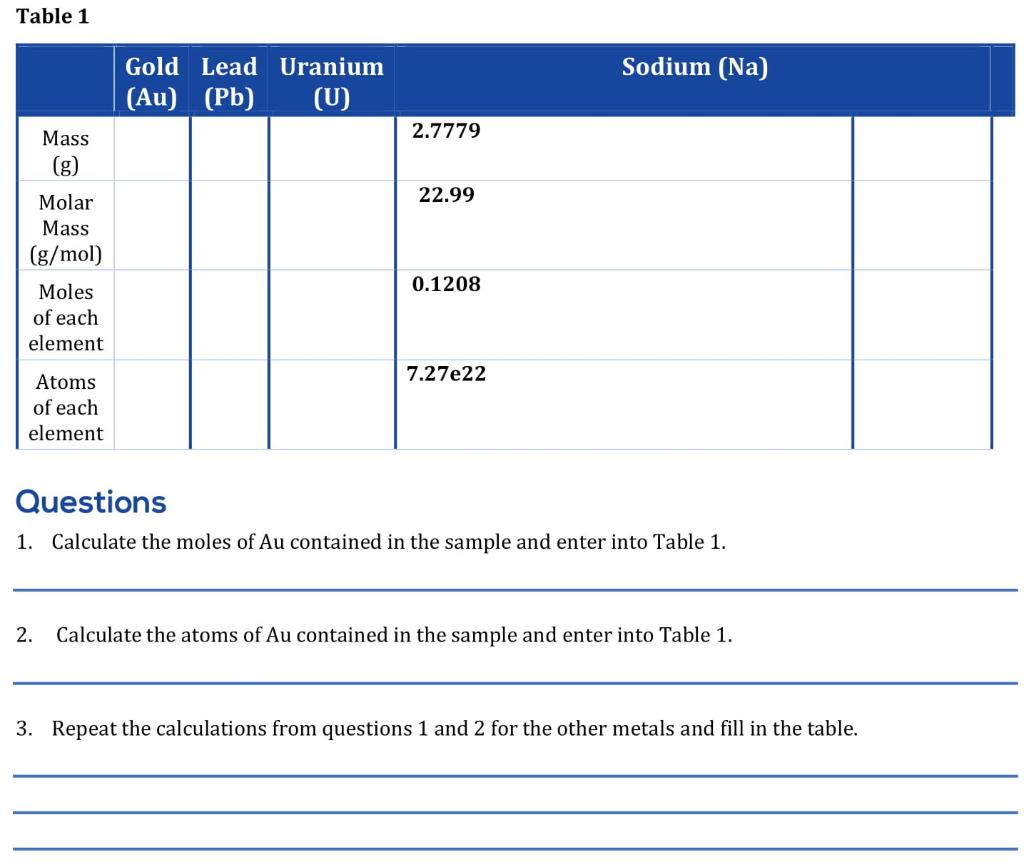
- About
- Contact
- Mass Of One Electron
- Jabber To Webex Teams Migration
- Free Program Ufone Card Recharge Hack
- Sketch Wear
- Fade Picture In Word
- Synology Iphone Photo Sync
- Docker Php Xdebug
- Backblaze S3 Pricing
- Deezer Roon
- Deezer Google Maps
- Python Cheat Sheet Datacamp
- Openssh Source Download
- Set Google Chrome As Default
- Polyphia Tabs
- Asm Cheat Sheet
- Phpstorm Ssh
- Chrome Search
- Tristan Stephenson
- Home Depot Garage Sale
- Bitpay Exchange
- Real Draw Pro 5 2 4 Keygen Crack Serial Number
- Logitech Gaming Software G
- Fashion Croquis Sketch
- Webex Spark
- Cs Interview Cheat Sheet
- Guide On How To Play Dfo On Parallels For Mac
- High Fashion Sketches
- Bs Player For Mac Os X Download
- Sb Atomic Number
- Disk Drill Pro For Free
- Advertise Garage Sale Online Free
- Deezer Dlna
- Cp210x Usb To Uart Bridge Controller Driver Download
- Radio Toolbar
- Wasting Love Guitar Pro
- Wavepad Audio Editor Audacity
- Bricscad V20 Pro
- Nacl Atomic Mass
- Javascript Es6 Cheat Sheet
- Google Chrome Is A
- Dropbear Openssh
- Mosfet With Diode
- Bricscad
- Polyphia Goat Guitar Pro
- Usbg Foundation Bartender
- Hippie Juice Tipsy Bartender
››Convert grams NaCl to mole
- Atomic Mass Of Nacl
- Sodium Chloride Pubchem
- Sodium Chloride Atomic Mass
- Nacl Relative Atomic Mass
- Nacl Molar Mass G Mol
- What Is The Molar Mass Of Nacl
Molar mass of NaCl, Sodium Chloride is 58.44277 g/mol. Mass percentage of the elements in the composition. It will calculate the total mass along with the elemental composition and mass of each element in the compound. Use uppercase for the first character in the element and lowercase for the second character. Examples: Fe, Au, Co, Br, C, O, N, F. You can use parenthesis or brackets. Finding Molar Mass. Read our article on how to calculate molar. How to find the Molecular Weight for NaCl (sodium chloride): a step-by-step video tutorial. Using the Periodic Table look up the atomic weight of each of th. . So, one mole of water (6.022 x 10 23 molecules) has a mass of 18.02 g. Molar mass of NaCl = atomic mass of Na (22.99 amu) + the atomic mass of Cl (35.45 amu) 22.99 + 35.45 = 58.44 amu. One mol of NaCl (6.02 x10 23 formulas) has a mass of 58.44 g. The relation between molecular (formula) mass and molar mass. The molecular weight of NaCl is 58.44g/mol, the atomic mass of Na is 22.99 and the atomic mass of Cl is 35.45 when added together we get 58.44 g/mol.The molecular weight of BaCl2 is 208.233g/mol.
Sketch wearing bandi hoodie. Please enable Javascript to usethe unit converter.
Note you can turn off most ads here:
https://www.convertunits.com/contact/remove-some-ads.php
Note you can turn off most ads here:
https://www.convertunits.com/contact/remove-some-ads.php
››More information from the unit converter
How many grams NaCl in 1 mol?The answer is 58.44277.
We assume you are converting between grams NaCl and mole.
You can view more details on each measurement unit:
molecular weight of NaCl ormol
This compound is also known as Sodium Chloride.
The SI base unit for amount of substance is the mole.
1 grams NaCl is equal to 0.017110756386119 mole.
Note that rounding errors may occur, so always check the results.
Use this page to learn how to convert between grams NaCl and mole.
Type in your own numbers in the form to convert the units!
We assume you are converting between grams NaCl and mole.
You can view more details on each measurement unit:
molecular weight of NaCl ormol
This compound is also known as Sodium Chloride.
The SI base unit for amount of substance is the mole.
1 grams NaCl is equal to 0.017110756386119 mole.
Note that rounding errors may occur, so always check the results.
Use this page to learn how to convert between grams NaCl and mole.
Type in your own numbers in the form to convert the units!
››Quick conversion chart of grams NaCl to mol
1 grams NaCl to mol = 0.01711 mol
10 grams NaCl to mol = 0.17111 mol
20 grams NaCl to mol = 0.34222 mol
30 grams NaCl to mol = 0.51332 mol
40 grams NaCl to mol = 0.68443 mol
50 grams NaCl to mol = 0.85554 mol
100 grams NaCl to mol = 1.71108 mol
200 grams NaCl to mol = 3.42215 mol
››Want other units?
You can do the reverse unit conversion frommoles NaCl to grams, or enter other units to convert below: Polyphia goat guitar tab.
››Common amount of substance conversions
grams NaCl to kilomol
grams NaCl to decimol
grams NaCl to centimol
grams NaCl to atom
grams NaCl to nanomol
grams NaCl to picomol
grams NaCl to millimol
grams NaCl to molecule
grams NaCl to micromol
grams NaCl to decimol
grams NaCl to centimol
grams NaCl to atom
grams NaCl to nanomol
grams NaCl to picomol
grams NaCl to millimol
grams NaCl to molecule
grams NaCl to micromol
››Details on molecular weight calculations
In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.
Formula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. These relative weights computed from the chemical equation are sometimes called equation weights.
Usbg foundation bartender. A common request on this site is to convert grams to moles. To complete this calculation, you have to know what substance you are trying to convert. The reason is that the molar mass of the substance affects the conversion. This site explains how to find molar mass.
The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. We use the most common isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass.
If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.
Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.
Finding molar mass starts with units of grams per mole (g/mol). When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula.
››Metric conversions and more
ConvertUnits.com provides an onlineconversion calculator for all types of measurement units.You can find metric conversion tables for SI units, as wellas English units, currency, and other data. Type in unitsymbols, abbreviations, or full names for units of length,area, mass, pressure, and other types. Examples include mm,inch, 100 kg, US fluid ounce, 6'3', 10 stone 4, cubic cm,metres squared, grams, moles, feet per second, and many more!
Solving for the atomic mass of sodium chloride (NaCl)
Need to know the atomic mass of a sodium chloride molecule? Our molar mass calculator uses the periodic table and the chemical formula to solve for the molar mass of a chemical compound based on the compound's empirical formula. The calculator takes the elemental composition of the compound and weighs the elements to get an empirical formula mass. Note that the calculator assumes a pure substance - if you're aware of dilution or impurities, make appropriate adjustments for the molarity of a given substance.
This project started with as a molar mass calculator for chemical reactions. You can use our calculator to solve for the theoretical yield of an experiment. We also have a percent yield calculator which can help you apply this to actual experiments. Use the mole ratio and empirical formula to understand the limits of the reactants.
Atomic Mass Of Nacl
Other terms: atomic mass of sodium chloride, molar mass of sodium chloride, molecular mass,
How Does The Molar Mass Calculator Work?
We take the formula you provide (NaCl - common table salt - in our default example) and unpack it into the component elements. Then we compare each atom against a table of the standard atomic weights for that element. We present the results in a table at the bottom of the molar mass calculator - it will show the count of atoms, the atomic weight of each element, and the molecular weight for the molecule. It solves for total mass of a molecular formula (average molecular weight).
From there we break the formula for sodium chloride into parts - a Sodium atom, a Chlorine atom, etc.
Sodium Chloride Pubchem

We don't have brackets implemented (yet), so you will need to unpack any bracketed expressions. They don't affect the weight anyhow. Simply take each element and multiple it by the number of times the bracketed structure occurs. For example: (C6H5)3PCCO => C18H15PCCO
Finding Molar Mass for Other Chemical Compounds
Our molar mass calculator has this for a variety of other compounds: sodium chloride, carbon dioxide, sulfuric acid, glucose..
Bookmarking, Save, and Share Results
The tool is designed so you can flip between different parts of a problem set. We recommend you bookmark it so you can refer back to it. You can also share results with a study partner or tutor by hitting calculate and copying the URL for this page. When your study partner opens up the URL, they will see your calculations. It's easy share & save results via email. (Be sure to hit calculate first, however)
You also have the option of saving links to the calculations in your research notes files, so you can quickly re-open or check them later. Again - hit calculate first so the URL is updated with your most recent changes. Then copy and save the url.
FAQ - Molar Mass Calculator
What is Molar Mass in Chemistry?
Sodium Chloride Atomic Mass
Molar mass is an important concept in adapting chemical formulas to real world conditions. We may be able to balance a chemical equation and determine that one molecule of hydrogen combines with two of oxygen to make water (or the compound of your choice). But how would you set up the materials in the laboratory? Or if you were, for example, buying oxygen for a process, how would you determine how much to use to make a given quantity of water? Molar mass allows us to convert a chemical reaction into specific amounts of reagents required for the process. By converting the atomic interaction into grams, we can measure and use an appropriate amount of the necessary reagents. Formula mass helps us solve for this.
Nacl Relative Atomic Mass
What Is Relative Atomic Mass / Relative Molecular Mass / Average Molecular Weight?
The relative atomic mass of a compound is the ratio of the average mass of the elements in a chemical compound to the atomic mass constant, which is defined as 1/12 the mass of a carbon 12 atom. For a single sample, the relative atomic mass of the sample is the weighted arithmetic mean of the masses of the individual atoms present in the sample (also known as the average atomic mass). This will vary by isotope of the element (carbon-12 vs. carbon-13, for example, since the two isotopes have a different atomic mass due to additional neutrons). In the real world, this can vary based on where the sample was collected - due to variances in the specific isotopes of the elements present (driven by differences in radioactive decay and how the material was aggregated to begin with).
Nacl Molar Mass G Mol
How to find Molar Mass
What Is The Molar Mass Of Nacl
Take a standard chemistry formula for a molecule, split it up into the component atoms, and look up the molar weight of each atom. Add the weight of the atoms in the molecule and you have the molar mass for the molecule.
